Maglern 60
A 9602 Thörl-Maglern
The LUKAS Heil-Betriebsstätte GmbH built at the Dreiländereck from Austria, Italy and Slovenia is a highly efficient production for steril and aseptic liquid.
learn more
The main shareholder of LUKAS Heil-Betriebsstätte GmbH is the LUKAS Foundation for Humane Medicine. The non-profit foundation supports scientific and medical research, the new or further development of natural remedies and studies in the field of innovative therapies.
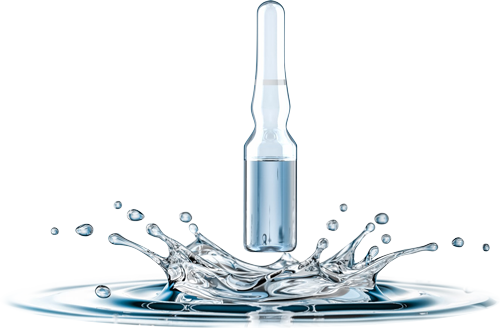
Due to the thermolability of the herbal raw material, the mistletoe preparation ISOREL® is aseptically ampouled – in compliance with the strict European GMP guidelines and under consideration of consistent complementary pharmaceutical criteria.
Free capacities are available for contract manufacturing and contract filling of aseptic or sterile drugs, food supplements and cosmetic active ingredients. Filling is in ampoule form, but also in vials.
The production rooms of LUKAS Heil-Betriebsstätte GmbH are equipped according to the latest pharmaceutical standards, including class A/B clean room areas with all the necessary facilities in accordance with the latest GMP (Good Manufacturing Practice) standards.
A visitor corridor surrounding the clean room, which is accessible at all times, allows potential contractors to easily convince themselves of the high quality of the production facility and processes and to carry out necessary audits.
As a self-sufficient Austrian company, we are able to react competently, reliably and flexibly to the requirements and developments of our time and thus offer our customers a real alternative to globalised production.
We would like to pass on our extensive know-how in the complementary medicine sector to our customers so that as many people as possible can benefit from the rhythms of nature.
This post is also available in: Ge